Isotope Substitution of Promiscuous Alcohol Dehydrogenase Reveals the Origin of Substrate Preference in the Transition State
View/
Impact
 Scholar |
Other documents of the author: Behiry, Enas; Ruiz-Pernía, José Javier; Luk, Louis Y. P.; Tuñón, Iñaki; Moliner, Vicent; Allemann, Rudolf K.
Scholar |
Other documents of the author: Behiry, Enas; Ruiz-Pernía, José Javier; Luk, Louis Y. P.; Tuñón, Iñaki; Moliner, Vicent; Allemann, Rudolf K.
Metadata
Show full item recordcomunitat-uji-handle:10234/9
comunitat-uji-handle2:10234/7013
comunitat-uji-handle3:10234/8638
comunitat-uji-handle4:
INVESTIGACIONMetadata
Title
Isotope Substitution of Promiscuous Alcohol Dehydrogenase Reveals the Origin of Substrate Preference in the Transition StateAuthor (s)
Date
2018-03-12Publisher
WileyISSN
1433-7851; 1521-3773Bibliographic citation
BEHIRY, Enas M., et al. Isotope Substitution of Promiscuous Alcohol Dehydrogenase Reveals the Origin of Substrate Preference in the Transition State. Angewandte Chemie, 2018, vol. 130, no 12, p. 3182-3185Type
info:eu-repo/semantics/articlePublisher version
https://onlinelibrary.wiley.com/doi/full/10.1002/ange.201712826Version
info:eu-repo/semantics/publishedVersionSubject
Abstract
The origin of substrate preference in promiscuous enzymes was investigated by enzyme isotope labelling of the alcohol dehydrogenase from Geobacillus stearothermophilus (BsADH). At physiological temperature, protein ... [+]
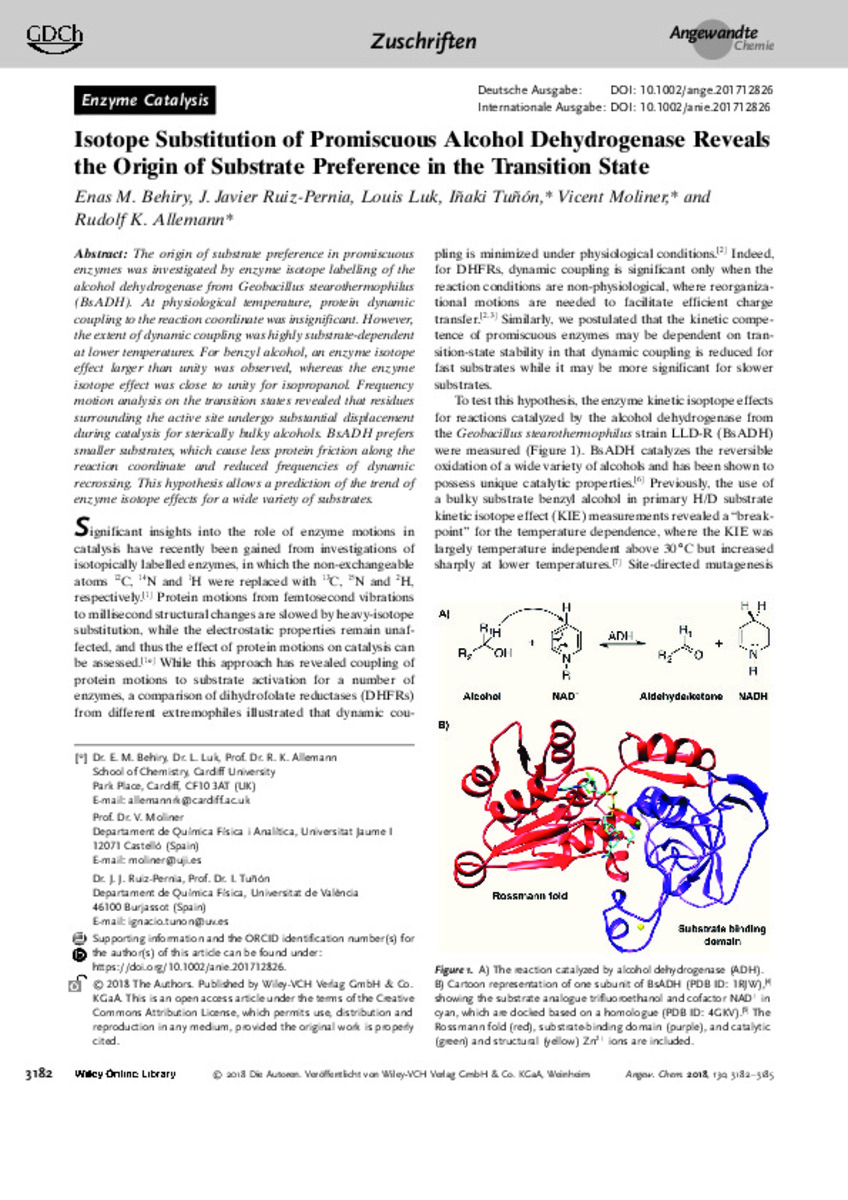
The origin of substrate preference in promiscuous enzymes was investigated by enzyme isotope labelling of the alcohol dehydrogenase from Geobacillus stearothermophilus (BsADH). At physiological temperature, protein dynamic coupling to the reaction coordinate was insignificant. However, the extent of dynamic coupling was highly substrate-dependent at lower temperatures. For benzyl alcohol, an enzyme isotope effect larger than unity was observed, whereas the enzyme isotope effect was close to unity for isopropanol. Frequency motion analysis on the transition states revealed that residues surrounding the active site undergo substantial displacement during catalysis for sterically bulky alcohols. BsADH prefers smaller substrates, which cause less protein friction along the reaction coordinate and reduced frequencies of dynamic recrossing. This hypothesis allows a prediction of the trend of enzyme isotope effects for a wide variety of substrates. [-]
Is part of
Angewandte Chemie, 2018, vol. 130, no 12Investigation project
Cardiff University through the UK's Biotechnology and Biological Sciences Research Council: BB/J005266/1, BB/L020394/1; Spanish Ministerio de Economia y Competitividad and FEDER funds: CTQ2015-66223-C2, CTQ2015-74523-JIN Universitat Jaume I: UJI.B2017-31; Generalitat Valenciana: PROMETEOII/2014/022Rights
info:eu-repo/semantics/openAccess
This item appears in the folowing collection(s)
- QFA_Articles [825]
The following license files are associated with this item:














